Accelerate Innovation and Compliance from Lab to Launch With Smarter User Adoption
Whatfix streamlines software adoption across life sciences—from R&D to commercialization. Boost productivity, simplify compliance with GxP-aligned workflows, and enable your teams to bring life-saving treatments to market faster.
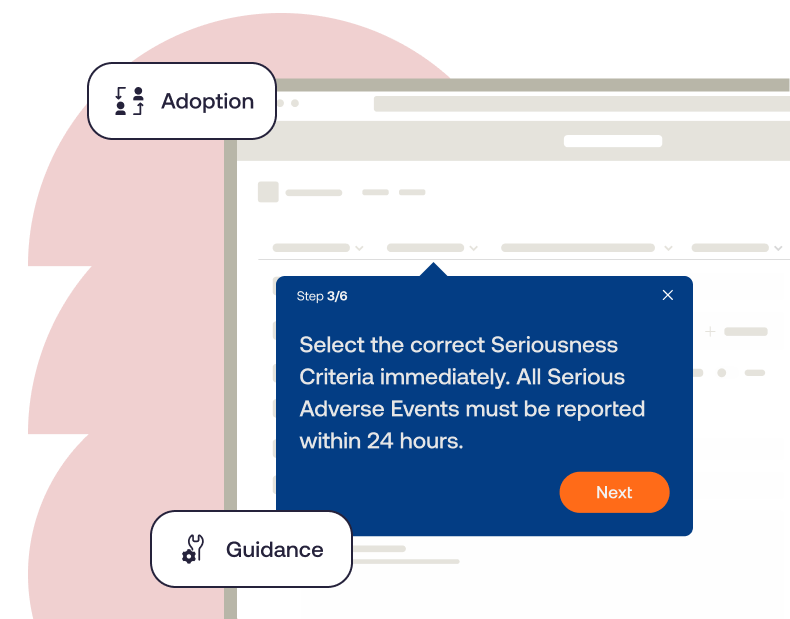
Enable Your Life Science Workforce With Guided Workflow Support
Support life science employees in the flow of work with role-based guidance and in-app assistance to drive efficiency and achieve business outcomes.
Simplify GxP Compliance with Ease
Ensure seamless adherence to evolving GMP, GCP, and GLP regulations. Whatfix simplifies compliance with in-app workflows, real-time alerts, and contextual guidance to reduce errors and improve confidence in your teams.
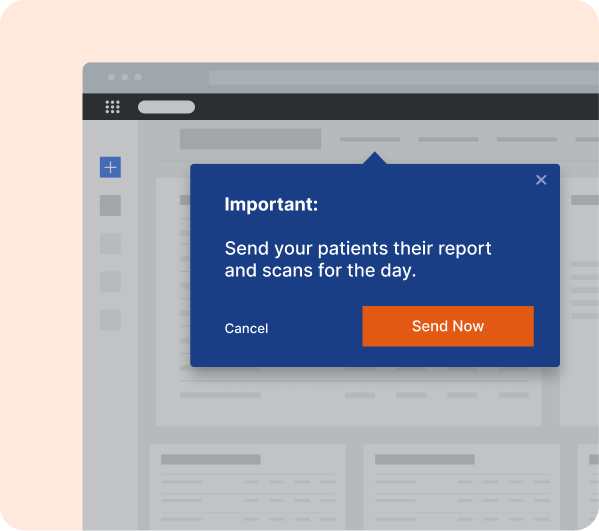
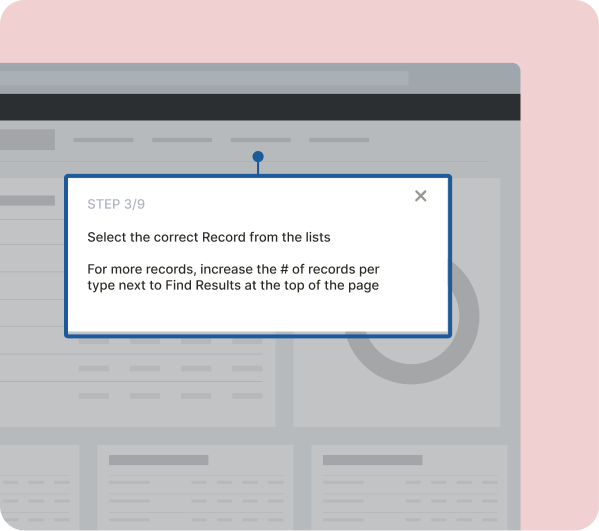
Faster Time to Market
Speed up clinical trials, regulatory approvals, and product launches with tailored in-app guidance, ensuring seamless processes and reduced delays.
Boost ROI and Operational Excellence
Transform productivity with user adoption tools that reduce training costs, streamline workflows, and drive better utilization of life sciences applications like Veeva and Salesforce. Your investment, maximized.
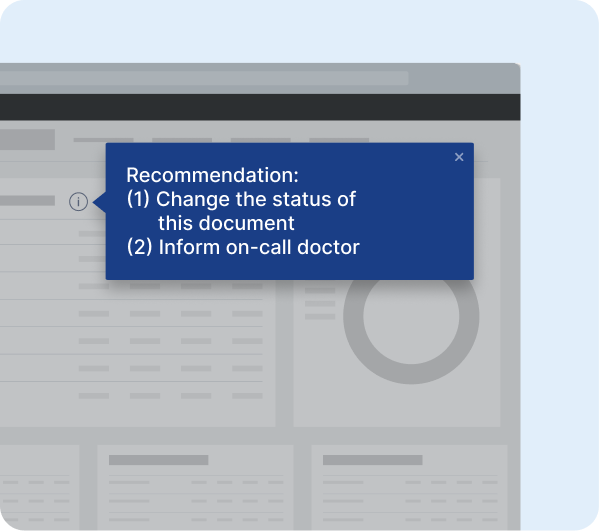
Unified platform to guide users and drive user adoption.
Pharma Software Clicks Better With Whatfix
See why Fortune 1000 pharma & life sciences companies choose Whatfix as their digital adoption partner.

Enterprise-Grade Security & Compliance
Whatfix adheres to the highest standards of information security, including ISO 27001, SOC 2 Type II, and GDPR compliance—ensuring your sensitive clinical, regulatory, and patient data remains secure.

Validated Learning Paths for Workflow Compliance
Build structured learning paths for different roles and regions to ensure SOP acknowledgment and compliance. Track completions and enable training teams to respond faster to audits and inspections.

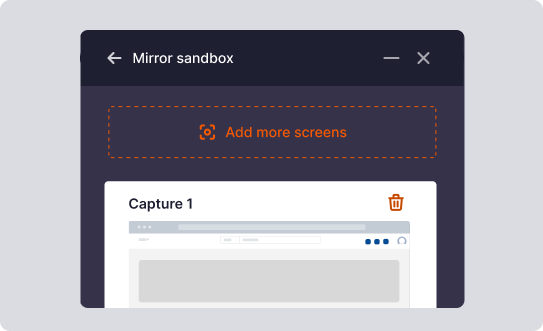
Simulation-Based Training
With Whatfix Mirror, create replica sandbox environments of your clinical, regulatory, quality, and more systems for hands-on training without risking live software usage.

Analyze and engage in one platform
Capture user events and usage data to identify areas of friction and take a data-driven approach to new in-app guidance, and improve life science experiences within a single platform.

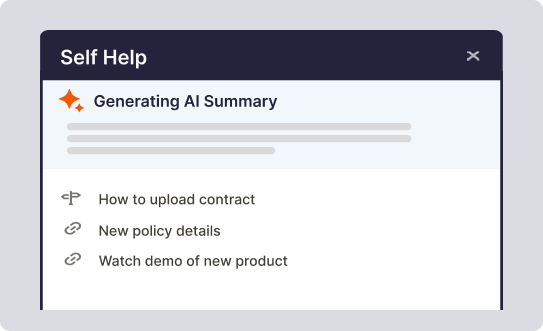
AI-powered adoption
Integrating generative AI into its digital adoption platform, Whatfix offers content authoring assistance, information synthesizing, performing in-app tasks based on user input, and more.

Customer-centric approach
Our digital adoption experts help you drive outcomes, from dedicated onboarding, post-deployment content author training, 24/5 support, our customer community, and Center of Excellence.
Trusted by Enterprises. Loved by Users.
Sheila Dusseau

33%
Reduction in contract workflow errors in three months.

WHITEPAPER
How DAP Enables Life Science Organizations to Achieve Full Technology ROI
Learn how embedding real-time, role-specific support directly within applications with a DAP enables life sciences teams to confidently use the tools critical to achieving life science innovation, regulatory readiness, and commercial success.
BLOG
Digital Transformation in Pharma
See how life science and pharma companies are driving business outcomes by transformation core business processes and operations – and how to accelerate digital ROI.
REPORT
The Forrester Wave™: Digital Adoption Platforms, Q4 2024
Read why Forrester named Whatfix a leader in its most recent Forrester Wave™ report.
BEST-IN CLASS SOLUTIONS
Recognized by
industry leaders

Software Clicks Better With Whatfix
In-app guidance. Hands-on simulations. Actionable analytics. One unified, AI-powered platform to drive adoption and maximize ROI.